|
Compared to 2007 and Office 2010 versions that have the tendency to crash. Office 2016 Visio Pro retail version activation key. In this case, it gives access to their definition, synonyms or translation from. Tags: microsoft office 2016 product key full version, office 2016 pro plus. Buy windows-7-professional-sp1 key. I can't update Microsoft office Professional Plus 2010 (32 bit) with Windows update. The MS office professional plus 2010 (32bit) was purchased under Microsoft @home use program, and the update was working fine until last few weeks. Microsoft office professional plus 2010 By Zepa (733.90 MB ) Microsoft office professional plus 2010 By Zepa (1.35GB ) Microsoft office 2010 professional plus with SP1 (1.18GB ) Microsoft office 2010 professional plus by pew (656.63 MB ) Microsoft office professional plus 2010 [Isohunt.to] (730.54 MB ) Microsoft office 2010 professional plus. (2) Sa bur prerak sarvniyanta, Updrashta bharta anumanta. Hanuman chalisa with meaning in gujarati pdf download. Silent mutations are being intensively studied. We previously showed that the estrogen receptor alpha Ala87’s synonymous polymorphism affects its functional properties. Whereas a link has been clearly established between the effect of silent mutations, tRNA abundance and protein folding in prokaryotes, this connection remains controversial in eukaryotic systems. Although a synonymous polymorphism can affect mRNA structure or the interaction with specific ligands, it seems that the relative frequencies of isoacceptor tRNAs could play a key role in the protein-folding process, possibly through modulation of translation kinetics. Conformational changes could be subtle but enough to cause alterations in solubility, proteolysis profiles, functional parameters or intracellular targeting. Interestingly, recent advances describe dramatic changes in the tRNA population associated with proliferation, differentiation or response to chemical, physical or biological stress. In addition, several reports reveal changes in tRNAs’ posttranscriptional modifications in different physiological or pathological conditions. In consequence, since changes in the cell state imply quantitative and/or qualitative changes in the tRNA pool, they could increase the likelihood of protein conformational variants, related to a particular codon usage during translation, with consequences of diverse significance. These observations emphasize the importance of genetic code flexibility in the co-translational protein-folding process. Introduction Nucleotide polymorphisms are DNA sequence variations that occur frequently within a population. Silent polymorphisms (those that do not change the amino acid in the encoded protein) have only in the last decade attracted increasing attention []. This kind of polymorphism can produce different effects on gene expression and lead to functional differences of diverse significance. Several recent reviews summarize the effects of such mutations, in particular in relation to human diseases, personalized biomedicine and pharmacogenomics [,,,,]. Silent mutations can affect gene expression acting at different levels and through different mechanisms. They can influence binding of regulatory factors on DNA, mRNA secondary structure and stability, ribosome traffic on mRNA and its interactions with specific ligands (as in riboswitches), including other RNAs and proteins. Silent mutations can also modify splicing, altering intron-exon boundaries or regulatory sites located in exons, enhancers or silencers, in all cases leading to an incorrect processing of mRNA. Finally, silent mutations can affect translational kinetics and protein folding, by changing codons read by tRNAs of different cellular availability. 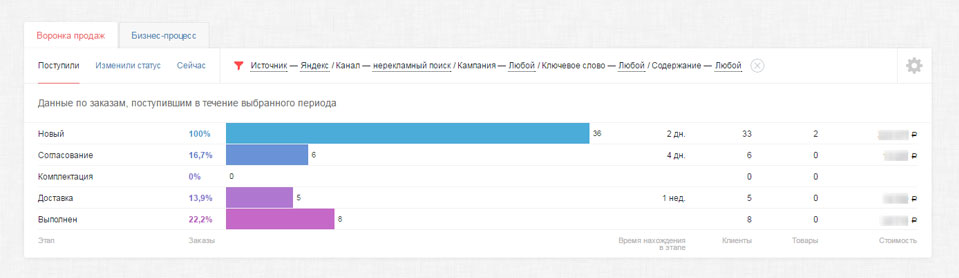
Therefore, through several mechanisms, silent mutations can give rise to differences in mRNA and protein abundance and in the structure and functionality of proteins (for a review see []). In this context, we have focused on the folding and biological activity of the human estrogen receptor alpha (hERα) in an attempt to understand whether silent mutations have any effect on the functional activity of this protein. Nearly 50 synonymous polymorphisms can be found in the hERα coding sequence []. Some of these have been further studied in order to establish an association with different human pathologies, most of them being associated with the risk of cancer development. Additionally, evidence points towards a role for some of these synonymous variants in sperm production and cognitive disorders. Two variants called PvuII and XbaI located in intron 1, or the (TA)n repeats in the 5’ UTR, are among the main polymorphisms studied in hERα []. These variants were found to be associated with various pathological conditions, including cardiovascular disorders, venous thromboembolism, miscarriage, and severe pre-eclampsia []. Recently, we studied the activity of the hERα variant ERAla87. This variant, assigned as Bst UI, is located in exon 1 and corresponds to an alanine codon change from GCG to GCC (rs746432). The mean GCC allele frequency was estimated in about 5%, varying between 0% and 10% in Asian and European populations, respectively. This synonymous variant has been studied for an association with several pathologies, and has only been associated with mood disorders, particularly in females []. Microsoft Office Professional Plus 2010 Product Key 
Using transiently transfected HepG2 and HeLa cells as an experimental approach, we were able to show that the functional activity of ERAla87 differs from that of the wild-type hERα, on a cell-type-dependent manner []. We propose that a conformational variant could be originated upon translation of ERAla87, as a consequence of differences in translational kinetics due to the availability of tRNA species that recognize either the GCG or the GCC codon. In this paper, we review arguments that support the idea that differences in the population of tRNAs could produce subtle changes in conformation of the ERAla87 and therefore explain the functional variations observed. Case of Study: ERαAla87 Synonymous Polymorphism hERα is a transcription factor which belongs to the nuclear receptor superfamily. It mediates, together with the estrogen receptor beta (ERβ), the pleiotropic and tissue-specific effects of estrogens. HERα presents a multidomain structure () which includes: the A/B domain containing the activation function-1 (AF1); the C-domain which holds the DNA-binding domain (DBD); a hinge region (D-domain); an E domain which harbors the ligand-binding domain (LBD), the dimerization interface and the activation function-2 (AF2); and the carboxy-terminal domain (the F-domain) [,]. Representation of the human estrogen receptor alpha functional domains (A to F). The location of activation functions 1 and 2, AF1 and AF2, are shown (above). 
Below, Estrogen receptor alpha (ERWT) and silent polymorphism ERAla87 coding sequences and translated amino acids residues around Ala87 are indicated. The nucleotide change in Ala87 is shown in bold. HERα can act through different mechanisms (). In the classical pathway, it binds directly to DNA, specifically to estrogen-response elements (EREs) located in promoters of estrogen-responsive genes []. In the non-classical genomic pathway, hERα interacts with other transcription factors (such as AP1 or Sp1) and regulates gene expression without directly binding to DNA [,,,]. HERα also acts via a “non-genomic” mechanism, in which it modulates the activity of kinases that can regulate gene transcription and the activity of other proteins [,]. HERα mediates estrogen (E2) and other ligand effects through three main pathways. (1) The classical pathway, in which hERα binds directly to DNA, to estrogen-response elements (EREs); (2) The non-classical genomic pathway, where hERα interacts with other transcription factors (e.g., AP1 or Sp1) and regulates gene expression without directly binding to DNA; (3) The “non-genomic” mechanism, in which hERα modulates the activity of kinases that can regulate gene transcription and protein activity []. Importantly, the hERα activation mechanism, the respective contributions of AF1 and AF2 towards its activity and the agonist/antagonist effect of different ligands and promoters are all cell-specific and depend on the differentiation stage of the cell [,,,,]. Installation motorisation portail battant somfy systems. For instance, HeLa cells, originated from a cervix carcinoma, present a poorly differentiated phenotype with a cell context strictly permissive to the AF2 transactivation function of hERα. In contrast, the hepatocarcinoma HepG2 cell line shows a more differentiated phenotype, and AF1 is the dominant transactivation function involved in hERα transcriptional activity in these cells [,]. Though important efforts have been made to understand hERα cell-specific activity and relevant advances have already been accomplished, to date the mechanisms underlying the modulation of its activity remain elusive. Differential Functional Properties of ERAla87 in HeLa and HepG2 Cells To explore whether the ERAla87 synonymous polymorphism presents a behavior similar to that of ERWT, we analyzed its transcriptional activity and subcellular localization comparatively to ERWT, in transfected HeLa and HepG2 cell lines.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
